|
4/17/2024 0 Comments Periodic table with charges ions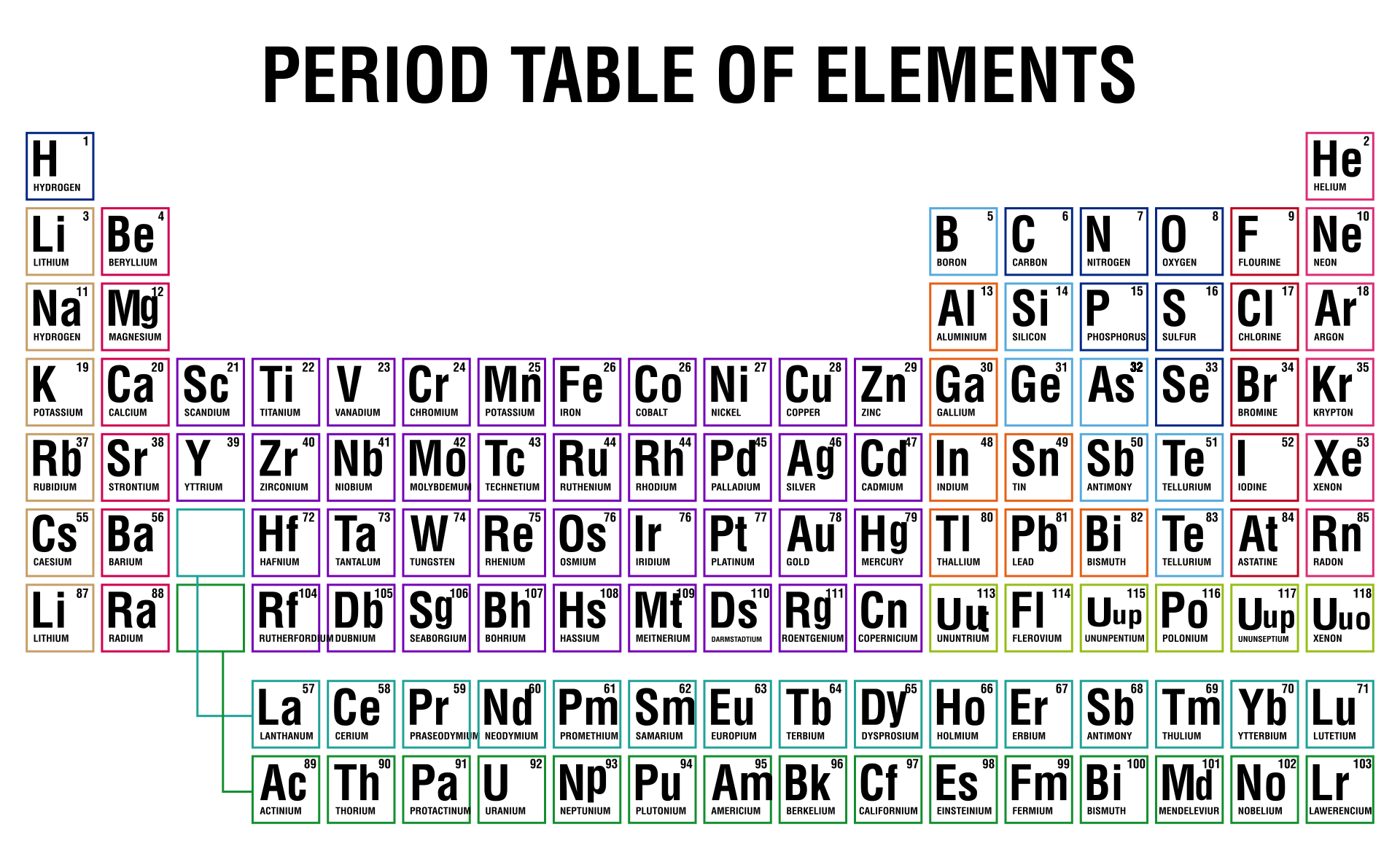
The chemical formula of magnesium chloride is MgCl 2. 
Similarly, when magnesium cations, Mg 2 +, combine with chloride anions, Cl −, there must be twice as many chloride anions as magnesium cations in order for the charges to balance each other. The formal charges are assumed to have only integer values. Note that charges do not appear in the chemical formulas of compounds: you don’t write Mg 2 +O 2 −). ionic model to be charged particles : ions Na +, Mg2 +, Al3 +, Ni2 +, F, O2-, etc. This reveals another corner chemists cut when writing formulas. Thus, when Mg 2 +and O 2 − combine to form the ionic compound magnesium oxide, there have to be equal numbers of magnesium cations and oxide anions. Learn what are ions and how they are formed by adding or removing electrons from atoms. And recall when oppositely charged ions combine to form compounds, there must be the same number of positive and negative charges, so that the compound carries no overall charge. For example, a sodium atom usually has a +1 charge, while oxygen most often has a -2 charge, and chlorine usually has a -1 charge. The valence of an atom is its oxidation state. 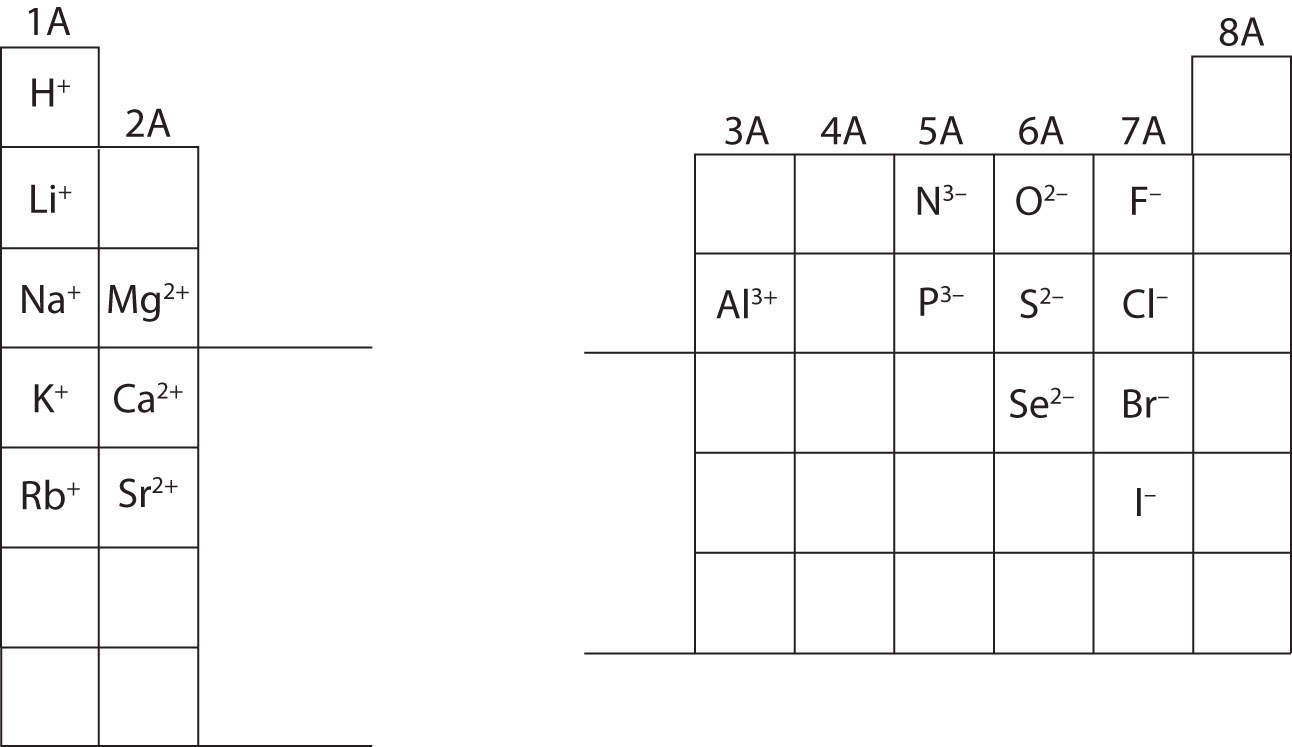
However, you can predict whether an atom will form negative or positive ions from its position in the Periodic Table: elements to the left (metals) will tend to form positive ions those on the right (non-metals) will tend to form negative ions. This printable periodic table contains the atomic number, element symbol, element name, atomic weights and most common valence charges. Note that the valency does not indicate whether an ion will carry a positive or negative charge: oxygen, O, also has a valency of two, and forms negative ions, O 2 −. Take for example, magnesium, Mg, this has a valency of two and tends to form ions carrying two positive charges, Mg 2+. In ionic compounds, valency gives an indication of the charge an ion formed from a particular element will carry. 3.3 Valency and predicting formulas in ionic compounds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
